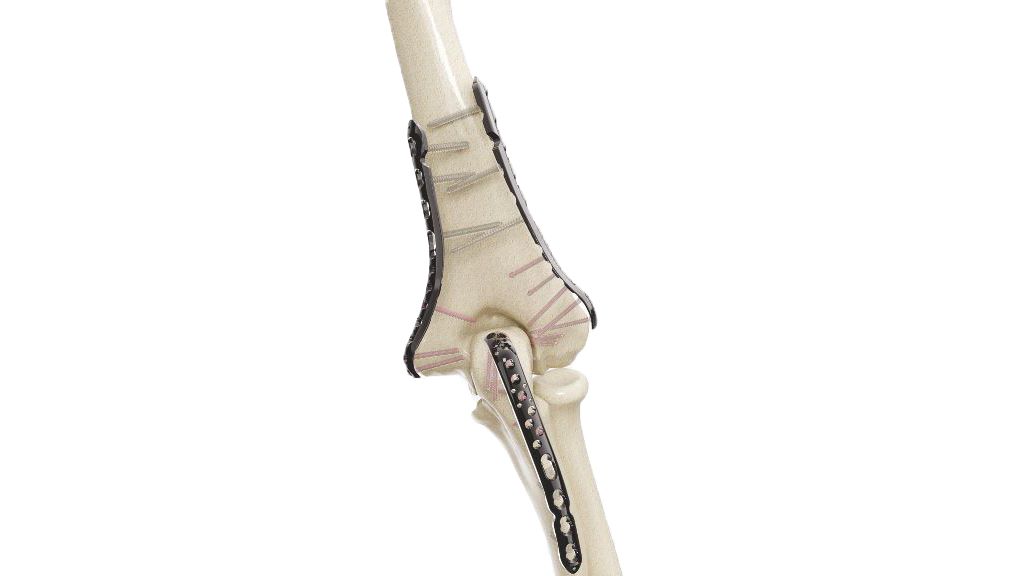
What goes in developing global quality
Orthopaedic implants & instruments?
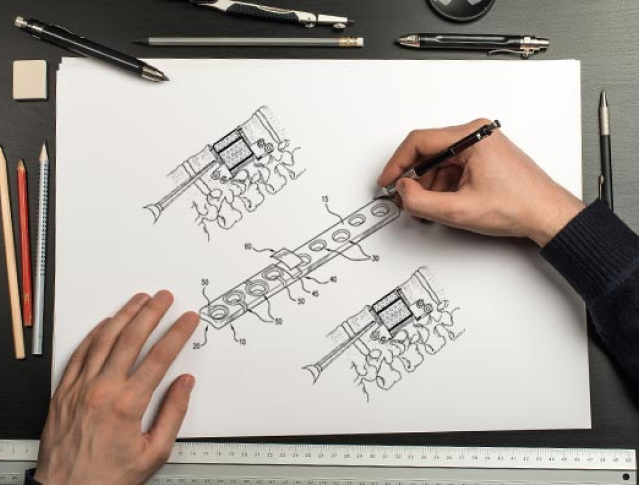
STEP 1CONCEPT
DEVELOPMENT
At Miraclus, we believe that the best products start with strong concepts which are not just a theory or model, but a “guiding principle”. Our concepts take birth at the problems our users face and go through the rigorous process of solution hypotheses, deliberation and risk assessment. Every aspect of the concept gets documented as the “Directive Principles Document”. The inimitable creativity of Miraclus’ engineers & technicians coupled with the wealth of experience & expertise provides unparalleled insights in our brainstorming and hands-on prototyping sessions. This enables us in rapidly developing the very best and simplest solutions through hand sketches, 3-D, CAD models and prototypes.
STEP 2DESIGN
FEASIBILITY
From Concept Development, we move to arrive at a “Steady Design” by evaluating the feasibility of the chosen concepts while considering all major project risks, including manufacturing obstacles and regulatory strategies. We achieve this by fully defining the design inputs (drawings, models &specifications), evaluating risks, and identifying and testing critical design features. We don’t move ahead until we are convinced of no major changes and have documented the bill of materials systematically, supported by an approved verification test plan.

STEP 3VERIFICATION &
VALIDATION
This is where we conduct design verification testing & analysis to make sure the product meets the defined requirements including functionality, strength, wear & fatigue. We also ensure that the risks are mitigated to the lowest possible level. Successful verification automatically moves to validation testing in a simulated environment ensuring that the product works as intended.

STEP 4LAUNCH
Our 40+ years of manufacturing experience provides us the in-depth knowledge & expertise to smoothly transition the design prototype into the shop floor (production unit) where our production engineers work seamlessly to make sure we deliver the product with its design intent.

.png)